COVID-19 Updates
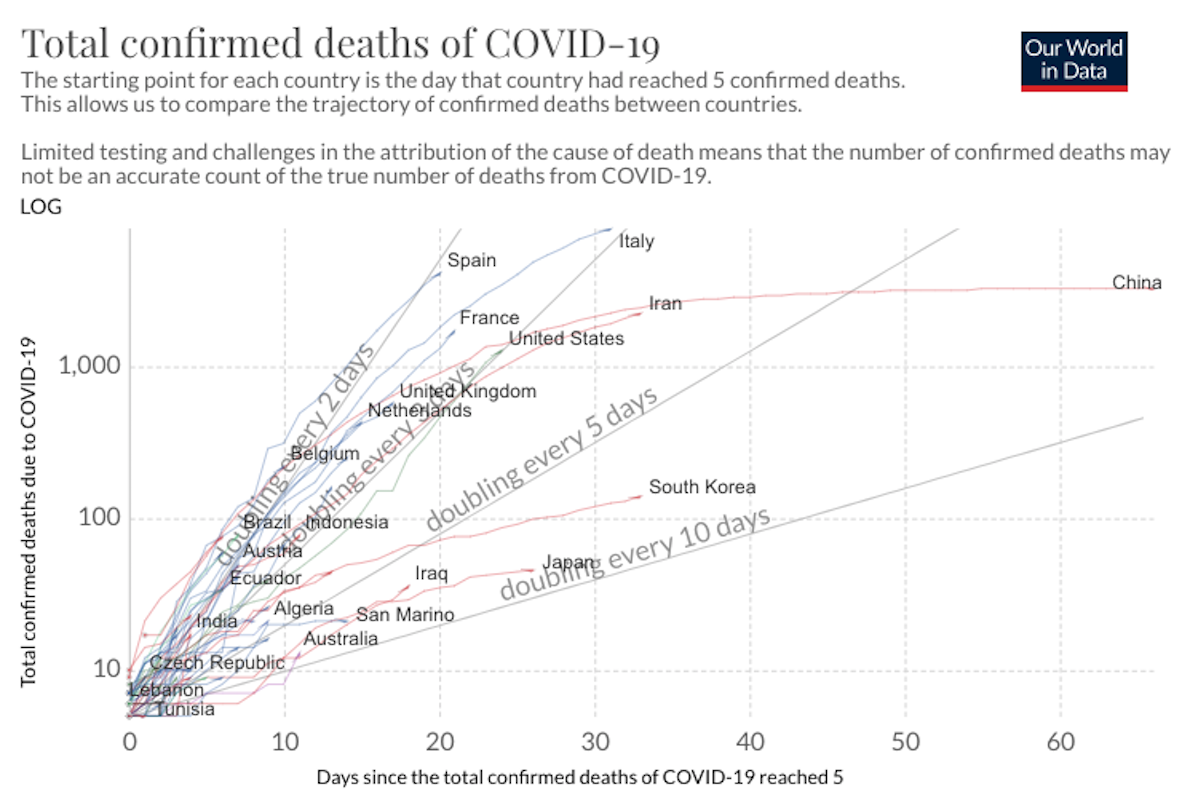
COVID-19 Science Update for March 27th: Super-Spreaders and the Need for New Prediction Models
Absent isolation or other precautionary measures, the average socially active COVID-19 infectee will transmit the disease to an average of about 2.4 people. i.e., the R0 value is 2.4. But super-spreaders can spread a disease to dozens or hundreds.
· 11 min read
Keep reading
Claire's Weekly Letter: Dangerous Losers
Claire Lehmann
· 3 min read
Nicholas Kristof and the Pornography of Accusation
Yuki Zeman
· 14 min read
Dangerous Losers
Jason Manning
· 10 min read
Frampton Comes Alive! at Fifty
Jacob Bielecki
· 13 min read