Science / Tech
How a UN Agency Misled the World on Cancer Risk
Nathan Schachtman’s indispensable new paper explains how the International Agency for Research on Cancer has misled the public with its classifications of carcinogens.
The International Agency for Research on Cancer (IARC) based in Lyon, France, is a highly regarded branch of the World Health Organisation. Its assessments and classifications of carcinogens are designed to alert the public as well as the cancer researchers, for whom such announcements are supposedly intended. However, the agency’s determinations have been the subject of increased scrutiny in recent years, and some of its findings have caused great controversy:
- In 2011, the IARC classified exposure to cell phones as “possibly carcinogenic to humans.”
- In March, 2015, the IARC classified glyphosate (the active ingredient in Roundup weedkiller) as “probably carcinogenic.”
- In October, 2015, the IARC classified red meat as “probably carcinogenic to humans” and processed meat as “carcinogenic to humans.”
While pronouncements like these elicited shock, confusion, and some ridicule from domain experts and the media, the IARC’s determinations remain influential, particularly in the United States, where they have sparked mass tort cases. Few people appreciate just how misleading the agency’s approach to classifying carcinogens is.

Researchers involved in the study of cancer and risk-communication—myself included—have commented on the IARC’s confusing edicts over the years (for instance, here, here, here, here, here, here, and here). What has been lacking is a thoroughgoing critical examination of the IARC monographs program, which identifies and classifies carcinogens—and its methodology. Now, 55 years after the agency began publishing its assessments, Nathan Schachtman—of counsel to UB Greensfelder and director of the Center for Truth in Science—has produced a tough-minded and statistically rigorous evaluation of the IARC’s methods, philosophy, and real-world results. His paper is a long-awaited corrective and a public service.
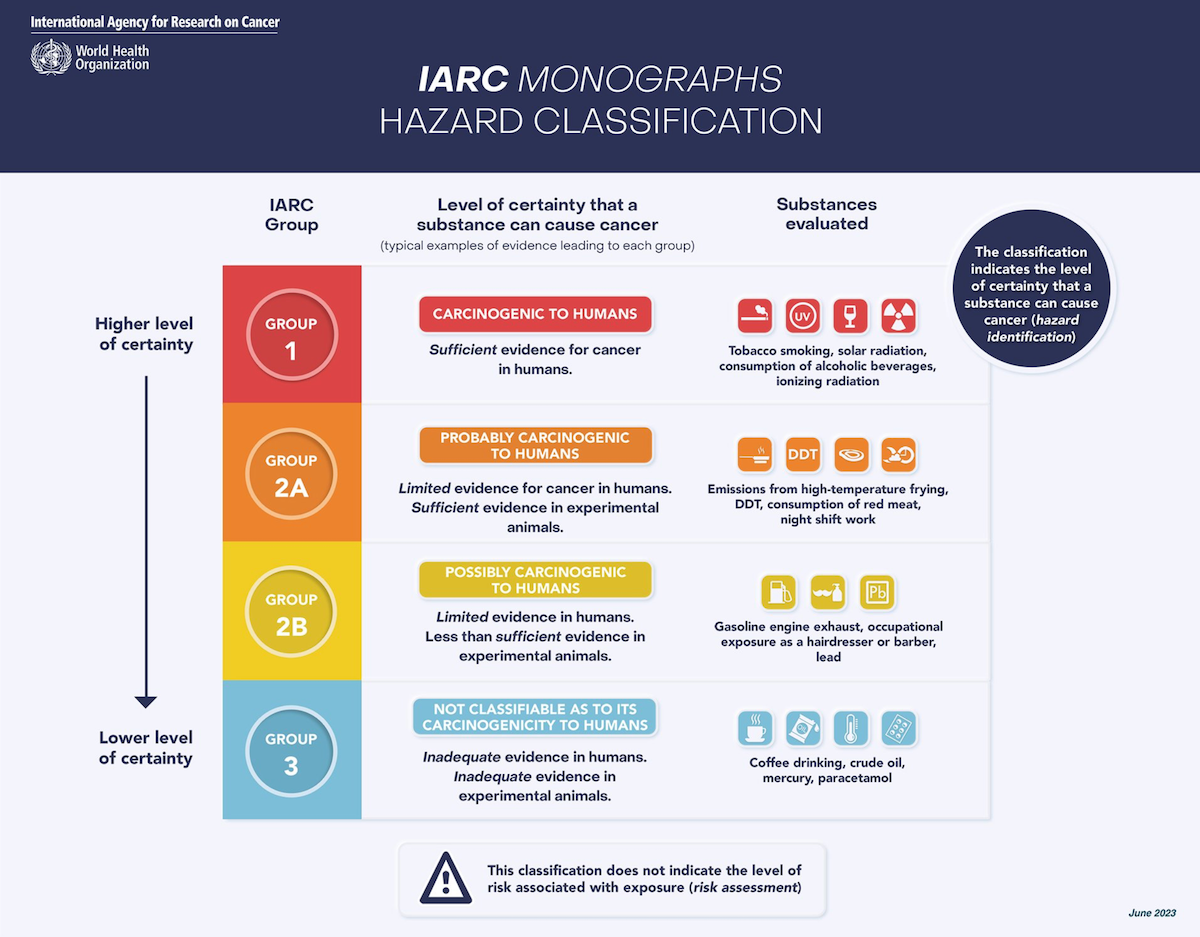
The IARC monographs use human epidemiological studies, animal experiments, and mechanistic studies using cell culture and bioassays to classify the carcinogenicity of agents in the following way:
- Group 1: Carcinogenic to humans (135 agents). Sufficient evidence for cancer in humans.
- Group 2A: Probably carcinogenic to humans (98 agents). Limited evidence for cancer in humans; sufficient evidence for cancer in experimental animals.
- Group 2B: Possibly carcinogenic to humans (326 agents). Limited evidence in humans; less than sufficient evidence for cancer in experimental animals.
- Group 3: Carcinogenicity to humans not classifiable (499 agents). Inadequate evidence in humans; inadequate evidence in experimental animals.
Data from human studies are the strongest kind. Animal experiments can show that a compound is carcinogenic in an animal species, but often this information may not be relevant to humans. Mechanistic studies can provide valuable information in combination with the other two types of data, but they cannot be decisive on their own.
The most important distinction in the IARC’s classification scheme is between agents in Group 1 (“known human carcinogens”) and those in Groups 2A, 2B, and 3. As Schachtman points out, although this carries the implication that substances in the remaining groups are not known to be carcinogens, the IARC suggests that substances in these other groups will likely or possibly prove to be carcinogens upon further investigation. The confusion caused by IARC’s classification scheme starts with these categories. Group 1 includes well-established agents like cigarette smoke, ionising radiation, alcohol, and the human papillomavirus, but also agents like processed meats and gasoline, for which the evidence of carcinogenicity is much weaker.
In Groups 2A and 2B, meanwhile, confusion is compounded by the agency’s idiosyncratic use of language. In the IARC’s usage, “probably” means something closer to “possibly” than “more likely than not.” Given this tendency towards grade inflation, Group 2B is sometimes assigned where the probability of carcinogenicity is even smaller than for Group 2A. As for Group 3—“not classifiable as to its carcinogenicity to humans”—Schachtman remarks that “IARC’s confusing definitions obscure the reality that group 3 agents are typically ones for which the evidence is insufficient, completely lacking, or even supportive of lack of carcinogenicity.”
The IARC’s rulings on coffee offer a good example of the low-information announcements in which the IARC specialises. In 1991, the agency labelled coffee a “possible carcinogen” based on “limited evidence” in humans—several case-control studies suggested that coffee drinking was associated with bladder cancer. However, the working group convened to evaluate coffee noted that, while a positive association was observed, factors like bias or the strong possibility of confounding (for example, by smoking—a strong risk factor for bladder cancer) could not be ruled out. In a 2016 evaluation, the IARC reclassified coffee as Group 3 (“not classifiable as to carcinogenicity”), a non-determination that ignores a wealth of evidence indicating that coffee is not carcinogenic in humans.
Going beyond the IARC’s elastic definitions of the four groups, Schachtman proceeds to expose the vague and, in some cases, difficult-to-distinguish terms used by the agency to characterise the existing evidence of carcinogenicity: “sufficient,” “limited,” “inadequate,” and “less than sufficient.” What, for example, constitutes “sufficient” evidence? And what happens when studies disagree—if, say, two find a strong association and others do not? In fact, he tells us that, “If there were ten animal studies, and only two studies found tumors in a single species, IARC consistency would be satisfied.”
Schachtman also finds that these terms are used differently when the intent is to acquit rather than indict, an imbalance that surfaces in the evaluation of all three types of evidence: human, animal, and mechanistic. For example, sufficient evidence of carcinogenicity in animals can be used to support the case for human carcinogenicity, but evidence suggesting a lack of animal carcinogenicity cannot be applied to humans.
Unlike other agencies, the IARC assesses hazard—that is, whether or not a substance is capable of causing cancer under certain conditions, such as feeding implausibly high doses to test animals or stimulating cell growth in a Petri dish. Other regulatory agencies usually assess risk—that is, whether or not exposure under real-world conditions affects the likelihood of developing cancer. It is significant that when the agency’s monographs program was established in 1971 to identify environmental carcinogens, the aim was to provide relevant data to regulatory scientists not broadcast information to the general public on how to prevent cancer.
Schachtman begins his investigation by reviewing the history of the IARC monographs program since its inception. The agency’s early focus on risk—principally that posed by chemicals—subsequently evolved into a focus on hazard that was broadened to include biological agents, lifestyle factors, and complex mixtures (tobacco smoke, air pollution). During this evolution, the IARC became increasingly wedded to the precautionary principle, which encourages researchers to err on the side of safety when an assessment of harm is doubtful. Schachtman’s critique of this now-ubiquitous principle is the lynchpin of his paper, illuminating the IARC’s determinations as well as its close links to activist organisations and the world of tort litigation:
The precautionary principle is rarely invoked with candid acknowledgments of ignorance. By asserting scientific conclusions not backed by sufficient valid evidence, precautionists undermine the scientific enterprise. By misrepresenting preliminary, incomplete, and poorly substantiated claims as supported by science, precautionists abridge and pretermit the important process of further scientific exploration.
The central thesis of this Monograph is that IARC has been captured by precautionary principle ideologues.
Schachtman’s critique of this problematic methodology entails analyses of specific determinations made by the agency; examination of conflicts-of-interest and the involvement of IARC scientists in tort litigation; and, finally, an evaluation of how the IARC’s methodology compares with the current standards for systematic literature review and meta-analysis.
The IARC’s espousal of the precautionary principle complements a belief that “all or most cancers are caused by man-made chemicals” and an inclination to accept evidence that is suggestive of harm, particularly from industrial products, such as cell phones, aspartame, glyphosate, DDT, talc, and hair dyes. At the same time, the IARC is so concerned with avoiding the appearance of industry influence that it excludes domain experts associated with industry from its working groups. This has led, on multiple occasions, to a distortion of the evidence weighed by those groups, the results of which are perhaps most evident in the agency’s notorious determinations about cell phones and glyphosate.
In the working group convened to evaluate cell phones, a highly qualified epidemiologist—who had conducted research on radiofrequency radiation—was dismissed from participation due to an alleged conflict of interest. Another epidemiologist—who had published research purporting to show an association of heavy cell-phone use with brain cancer, and who had testified in court cases on the question—was allowed to play a prominent role in the working group. Furthermore, the “possible carcinogen” determination was based primarily on what the working group itself considered “limited evidence” from two case-control studies, whereas data from a large prospective study, which showed no association, was ignored, along with extensive “ecologic” data from many countries showing no increase in brain-cancer rates in the two decades following the introduction of cell phones.

The IARC’s assessment of the herbicide glyphosate, published in March 2015, is even more problematic. Glyphosate has been in use since 1974, and it remains the most popular weedkiller worldwide. Twenty-two national and international health agencies have found it to be safe and not carcinogenic at the levels to which most people are exposed. The IARC’s determination is at odds with the findings of all of these other agencies. Furthermore, scrutiny of the IARC’s process in evaluating glyphosate has revealed improprieties, including statistical errors, selective editing, and conflicts-of-interest.
Schachtman reminds us that industry-funded research has long been distrusted, while research funded by environmental or advocacy groups—or by government departments like the National Institute for Environmental Health Sciences—is presumed to be free of bias. Since the 1960s and 1970s, the toxic tort and carcinogen-litigation industry has grown from a niche area into one of the fastest-expanding segments of the US legal system. For over three decades, the IARC has had a close connection with the Collegium Ramazzini, an institution founded in 1982 to be an independent “academy” concerned with occupational and environmental health. Many members and fellows of the Collegium serve on IARC panels and have worked regularly with US tort law firms as litigation consultants and expert witnesses, especially in cases involving chemicals like glyphosate, benzene, asbestos, and talc.
The intimate relationship between the IARC’s classification methodology and the activities of its scientists in the litigation sphere is evident in a new tool that the IARC uses to increase its ability to detect carcinogenicity. In 2016, Ramazzini fellow Martyn Smith of the University of California at Berkeley published a checklist of ten key characteristics of carcinogens, and this list was subsequently incorporated into the IARC’s guidelines. The characteristics include whether or not an agent is electrophilic, is genotoxic, alters DNA, induces epigenetic alterations, induces oxidative stress, and so on. As with the IARC’s consideration of human, animal, and mechanistic evidence, there is no requirement for specifying the intensity or duration of these conditions, and a study evaluating these key characteristics found that they were no better at identifying carcinogens than chance. As David Zaruk—an expert in environmental regulation—has commented, “Anything that had been previously determined by regulatory risk assessments to be non-carcinogenic (like glyphosate, gasoline or aspartame), could now, under this new Ramazzini standard, be concluded to be a carcinogen.”
Throughout his paper, Schachtman draws attention to the many inventive ways in which the IARC skews the interpretation of the evidence, and not just by leveraging ambiguous terms and dubious checklists. Longstanding principles of toxicology and epidemiology are also conspicuously absent from the IARC’s assessments. The cardinal principle of toxicology is that “the dose makes the poison,” but when alerting researchers and the public to the potential carcinogenicity of a given agent, the IARC does not mention that there can be thresholds and benefits at different levels of exposure. Also absent from the IARC’s thinking is the work of Bruce Ames, which shows that we are exposed to much higher levels of naturally occurring pesticides from eating broccoli and cabbage than we are to synthetic pollutants and carcinogens. Finally, the IARC pays scant attention to Sir Austin Bradford Hill’s 1965 “considerations” when assessing causality.
The IARC’s determinations regarding carcinogens have been subject to criticism since at least 2008, when a paper by Boffetta et al titled “False-Positive Results in Cancer Epidemiology: A Plea for Epistemological Modesty” was published. The authors emphasised the IARC’s tendency to be misled by “false positives”—initial results that appear to show a risk but that fail to replicate in larger and more careful studies. In this and other papers (here, here, and here), critics raised questions about the IARC’s criteria for identifying carcinogens, about specific determinations, about the make-up of working groups and potential conflicts-of-interest, and about the extreme secrecy surrounding deliberations.
In February 2015, the IARC published a paper in which 126 authors, led by epidemiologist Neil Pearce, responded to the agency’s critics and declared their concerns “unconvincing.” Most striking in this response is how unwilling the IARC scientists were to engage with substantive criticisms of the agency’s classification methods and the decisions it produced. Basically, the IARC’s response to criticism is to imply—or state outright—that critics are biased by connections to industry, and that a review of IARC practices confirms that nothing needs to be modified.
But Schachtman finds that the IARC’s insulation from practices followed by other scientific and regulatory agencies extends to its conduct of systematic reviews and meta-analyses. Before the 1990s, there were few agreed-upon standards for reviews of the literature in the area of carcinogenesis. In this environment, authors tended to rely on their subjective judgment to determine which studies merited attention, what their results showed, and how consistent the evidence was. Over the past thirty years, however, standards for synthesising evidence in the area of cancer causation have become much more sophisticated and the quality of systematic reviews has greatly improved. The US EPA and OSHA have developed their own approaches to classifying carcinogens in the environment and the workplace that reflect these changes, but the IARC has not kept pace with these developments.
The IARC’s enormous influence on the classification of carcinogens and on tort litigation in the US has been responsible for tens of billions of dollars in damages. At the same time, its determinations of carcinogenicity have been the subject of increasing scrutiny and criticism. Schachtman concludes his analysis by arguing that there is no justification for the US government to continue to support the IARC’s monographs program. However, he is also careful to point out that other divisions of the agency perform valuable functions, including tabulating cancer statistics from different countries, standardising pathology codes, and carrying out original international research programs.
For the past thirty years, for instance, Nubia Muñoz and colleagues have conducted landmark molecular epidemiological studies across different countries into human papillomavirus (HPV) infection and cervical cancer, as well as oropharyngeal and other cancers. This work, which was conducted away from the limelight, demonstrated the role of HPV as the central and necessary cause of cervical cancer and contributed to the groundwork for the development of a vaccine. But successes like these only serve to illuminate the failings of the monographs program by comparison—particularly its closed-mindedness and resistance to criticism. If the best work of the IARC is to be protected, its flaws must also be urgently addressed.
Quillette invites thoughtful responses to its essays.
Selected responses are published once per week as part of a curated Letters to the Editor feature. If selected, letters appear under the contributor’s real name and may be edited for clarity and length.
To submit a letter for consideration, please email [email protected].






